Jack Fox
Biophysical Characterization of Apo and Holo Riboflavin Binding Protein
Abstract:
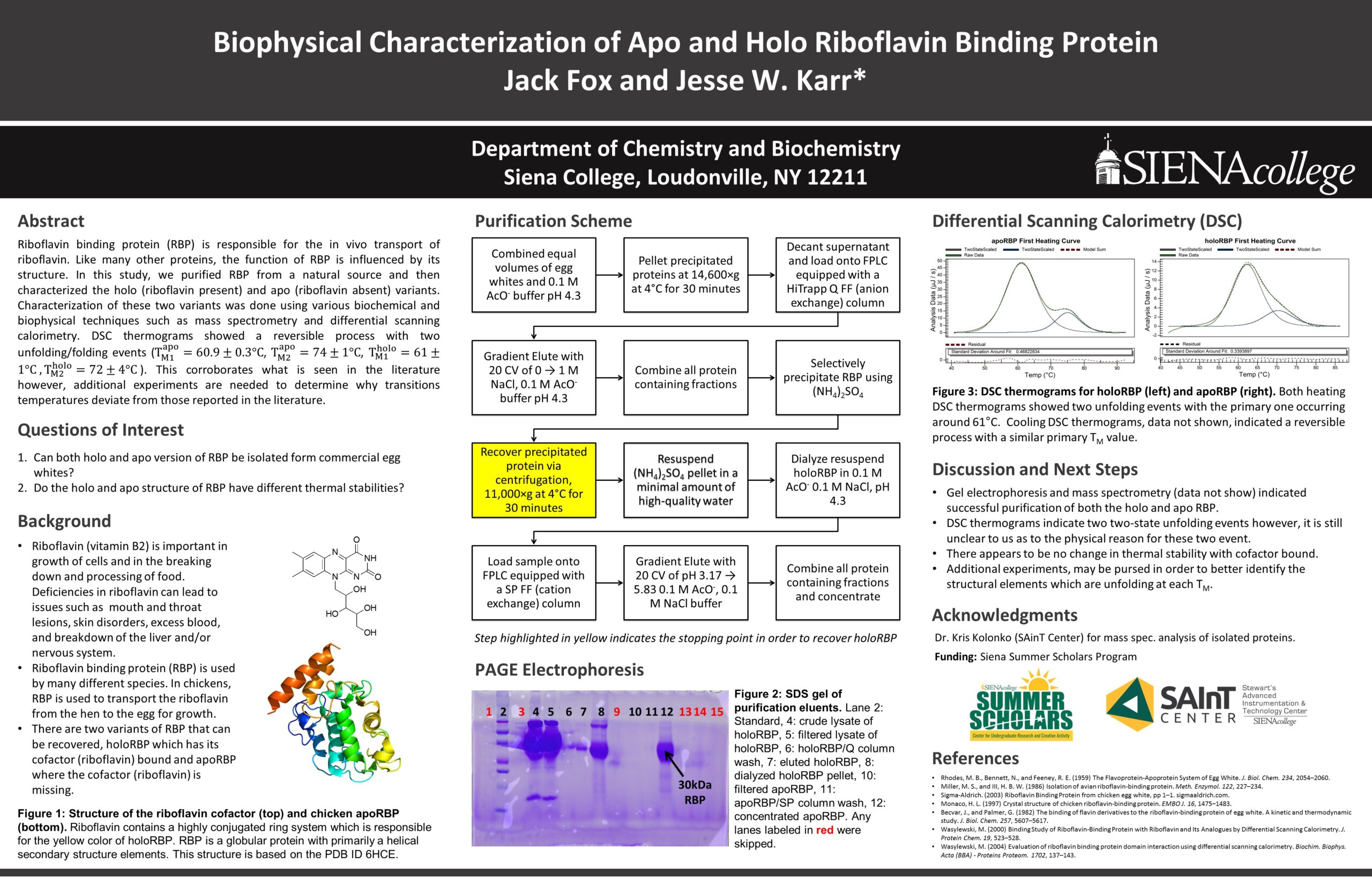
Riboflavin binding protein (RBP) is responsible for the in vivo transport of riboflavin. Like many other proteins, the function of RBP is influenced by its structure. In this study, we purified RBP from a natural source and then characterized the holo (riboflavin present) and apo (riboflavin absent) variants. Characterization of these two variants was done using various biochemical and biophysical techniques such as mass spectrometry and differential scanning calorimetry. DSC thermograms showed a reversible process with two unfolding/folding events (T_M1^apo=60.9±0.3°C, T_M2^apo=74±1°C, T_M1^holo=61±1°C ,T_M2^holo=72±4°C). This corroborates what is seen in the literature however, additional experiments are needed to determine why transitions temperatures deviate from those reported in the literature.Title
Biophysical Characterization of Apo and Holo Riboflavin Binding Protein
Faculty Advisor
Dr. Jesse Karr
Location
Table 42

